
EUROLAB BIOCONSULTING
euroLAB Bioconsulting is mainly dedicated to Biosafety, specializing in the inspection of Indoor Air Quality, validation of controlled environment rooms and Biodescontamination of critical areas.

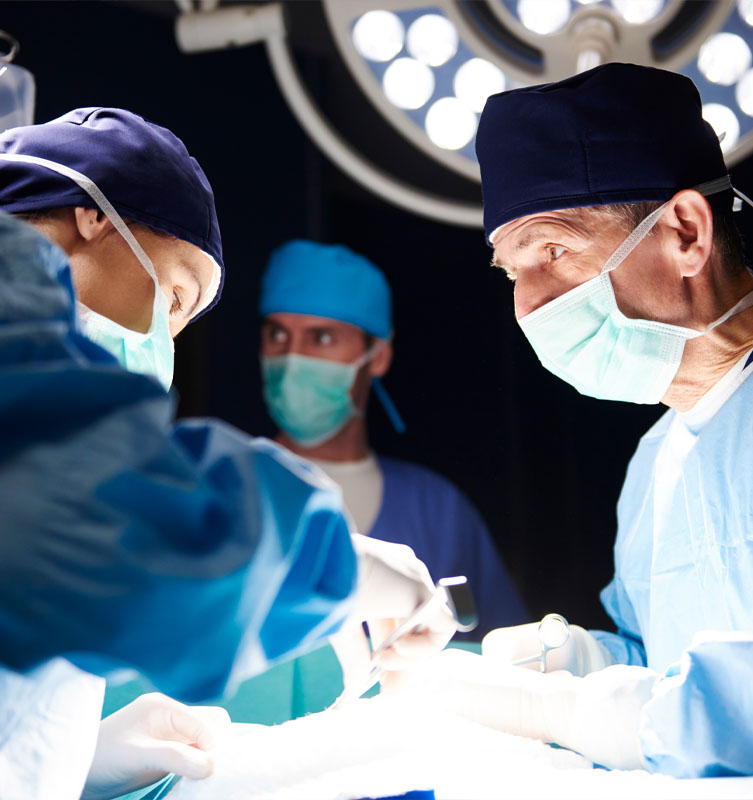

Validation of critical areas in hospitals
To avoid nosocomial infections, validation of critical areas is vital.
Hospitals are complex buildings that require special control and even more so in their operating rooms, laboratories, ICU, etc. Annual validation of environmental and biosafety conditions must be carried out in these rooms.
GMP validation of clean rooms and laboratories
The manufacture of sterile products is subject to special requirements to minimize the risks of microbial, particulate and pyrogen contamination.
euroLAB has the best technology for the validation of pharmacy laboratories, since it has high flow particle counters that allow shortening particle counting times.










